No products in the cart.
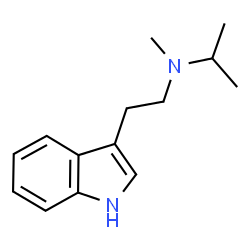
MiPT
€10.15
Buy Clean and pure MiPT [>93.0%] direct from our online shop
Shipping to USA, EU, NZ, AU *
*for research purposes only. human consumption is against our terms of service.
like with most psychedelic tryptamines, mipt is thought to act principally as a 5-ht2a partial agonist. the psychedelic effects are believed to come from mipt’s binding efficacy at the 5-ht2a receptors.
| bulk discount: | |
| 1 Gram | 10.15 |
| 3 Grams | 23.00 |
| 5 Grams | 31.50 |
| 10 Grams | 61.00 |
| 25 Grams | 139.50 |
| 50 Grams | 254.00 |














HarmB –